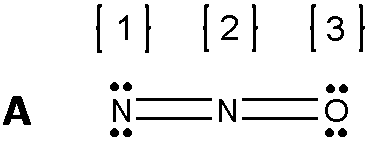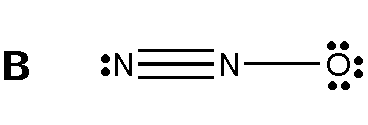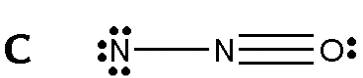From chemistry class, you may have already been familiar with what a Lewis Structure diagram looks like for specific molecules. However, you may also have noticed that there are more than just one possible Lewis Structure for some molecule/compound. Thus, before we can understand N2O's molecular structure, we must be familiar with the term resonance.
ResonanceResonance Structures appears when there are two or more Lewis Structure of a molecule that have the same skeleton but different distribution of electrons around the structure. It is used when one Lewis Structure is not sufficient in illustrating the bonds of a molecule.
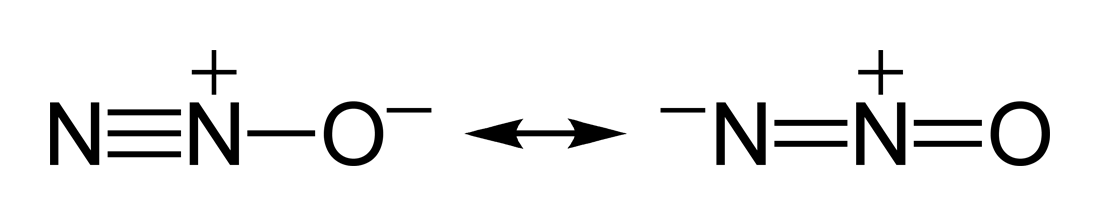
But why does this happen? Why can't there be just one definite Lewis structure for one molecule? To answer this, we must understand that electrons, unlike the nucleus have no fixed position within the structure. Rather, the Lewis diagram illustrates the highest probability of the general space an electron will fill. These electrons will move around the molecule to stabilize the compound, and for some compounds this would result in a different distribution of electrons that would result in different versions of a Lewis structure. In the case of Nitrous Oxide, there are two major resonance structures as illustrated by Figure 2. |
Figure 2. Resonance structure of N2O [Digital Image]. Accessed April 11th. 2016. Retrieved from http://www.carewelldentistry.com/services-laughing-gas/
|
Deriving Resonance Structure using the Octet rule and Formal charge
|
Figure 3. Formal Charge Formula [Digital Image]. Accessed April 11th, 2016. Retrieved from http://www.chem.ucalgary.ca/courses/351/Carey5th/Ch01/ch1-3-2.html
Formal charge; |
When deriving a resonance structure, we must consider the octet rule as well as formal charges.
You may already be familiar with the octet rule, in which the element associated within the molecule should have a full valence shell. But what exactly is a formal charge? |
Formal charge refers to the specific charge of an atom within the molecule and is calculated by the formula illustrated in Figure 3. Formal charge can be used as a tool to assess a resonance structure, and it assumes the atoms in the molecule have equal electronegativity.
- It is important to note that there are 2 electrons within a bond or a "line" in a Lewis structure, when counting individual electrons for the calculation
Applying this to Nitrous oxide
All of these Lewis structure fulfills the octet rule, but to figure out which structures are ideal, we must consider formal charges.
|
Formula; (# of valence electron) - (# of covalent bond) - ( # of lone electrons)
1 N; 5 - 2 - 4 = (-1) [Negative charge] 2 N; 5 - 4 - 0 = (+1) [Positive charge] 3 O; 6 - 2 - 4 = 0 [Neutral] From this calculation, the formal charge illustrates that the net formal charge on the terminal Nitrogen (-1) and the middle nitrogen (+1) have a net formal charge that equals to the overall charge of the molecule which is zero. Thus, a possible resonance structure. |
|
Formula; (# of valence electron) - (# of covalent bond) - ( # of lone electrons)
1 N; 5 - 3 - 2 = 0 [Neutral] 2 N; 5 - 4 - 0 = (+1) [Positive charge] 3 O; 6 - 1 - 6 = (-1) [Negative Charge] From this calculation, the net formal charge also equals to the overall charge of the molecule, as Oxygen have a negative charge while being bonded to a Nitrogen with a positive charge. Making this a possible structure. |
|
Formula; (# of valence electron) - (# of covalent bond) - ( # of lone electrons)
1 N; 5 - 1 - 6 = (-2) [Negative Charge] 2 N; 5 - 4 - 0 = (+1) [Positive Charge] 3 O; 6 - 3 - 2 = (+1) [Positive Charge] From the looks of this calculation, the net formal charge of each structure in the molecule is higher than the overall charge of the molecule. The Terminal Nitrogen Charge is 2- while the Nitrogen bonded to it is only 1+. Thus, this resonance structure is not likely to occur, and therefore not a structure that we would usually consider. |
Figure 4. N2O Lewis structure [Digital Image]. Accessed April 11th, 2016. Retrieved from http://www.chegg.com/homework-help/questions-and-answers/structure-formal-charges-atoms-1-2-3-structure-b-formal-charges-atoms-1-2-3-structure-c-fo-q1174255
Using the structure to explain N2O's polarity
Now that we have established why the molecule for Nitrous oxide exists in different forms, switching from one structure to another for stability, we can now understand why this molecule have a low polarity.
In the previous page we mentioned how the charges cancels each other out which results in a low polarity.
In Figure 5, the resonance structures contributes a negative formal charge at each end of the molecule. One on the terminal Nitrogen and the other on the terminal Oxygen, since each end is negatively charged and tends to attract electrons in the opposite direction of each other, the result is a cancellation of those attraction force to a certain degree. Keep in mind that there are also electro-negativity differences to consider.
In the previous page we mentioned how the charges cancels each other out which results in a low polarity.
In Figure 5, the resonance structures contributes a negative formal charge at each end of the molecule. One on the terminal Nitrogen and the other on the terminal Oxygen, since each end is negatively charged and tends to attract electrons in the opposite direction of each other, the result is a cancellation of those attraction force to a certain degree. Keep in mind that there are also electro-negativity differences to consider.
Figure 5. Major Resonance Structure of N2O [Digital Image]. Accessed April 11th, 2016. Retrieved from http://www.senseair.com/senseair/gases-applications/dinitrogen-oxide/